Sep 22 2015
Scientists from the Max Planck Institute for Polymer Research (MPI-P) in Mainz, Germany and FOM Institute AMOLF in the Netherlands have collaborated to discover that the structural memory of water exists on a picosecond timescale.
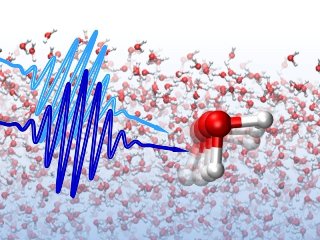 The lifetime of local water structures is probed using ultrafast laser pulses. Credit: © Yuki Nagata / MPI-P
The lifetime of local water structures is probed using ultrafast laser pulses. Credit: © Yuki Nagata / MPI-P
The team utilized innovative ultrafast vibrational spectroscopies for characterizing the local structural dynamics that exist in liquid water.
The dynamics could be explained as the speed with which water molecules alter their binding state, and it has been observed that local structures maintain their structural memory for more than a picosecond.
Water has generally been considered as a solvent and the observations made in this study change that perception. “71% of the earth’s surface is covered with water. As most chemical and biological reactions on earth occur in water or at the air water interface in oceans or in clouds, the details of how water behaves at the molecular level are crucial. Our results show that water cannot be treated as a continuum, but that specific local structures exist and are likely very important” says Mischa Bonn, director at the MPI-P.
Water has ultra fast dynamics. On sub-picosecond timescales, water makes various movements like jiggling and wiggling, and on such a short timescale, they cannot be clearly distinguished. The molecules of water are considered to lose their memory about their local structure within an ultra short time period of less than 0.1ps.
The team employed ultrafast infrared spectroscopy to measure Oxygen-Hydrogen (O-H) bond vibrations in water. They concentrated on water molecules that were strongly and weakly hydrogen-bonded to the water molecules nearby. They observed that when the water molecules had a large separation, the vibrations lived longer to about 1ps, and when the water molecules were close, the vibrations lived to just 0.2ps. The team had found that water molecules that were weakly bound continued to remain weakly bound for a longer time period.
In this study, the researchers have shown the uniqueness of liquid water when compared to other molecular liquids.
The study paper has been published in the journal, Nature Communications.