 By Surbhi JainReviewed by Susha Cheriyedath, M.Sc.Aug 25 2022
By Surbhi JainReviewed by Susha Cheriyedath, M.Sc.Aug 25 2022In an article recently published in the open-access journal Materials, researchers discussed the development of films made from biopolymers that are more environmentally friendly and have improved photo-oxidative resistance.

Study: Ecofriendly Biopolyester-Based Nanocomposite Films with Improved Photo-Oxidative Resistance. Image Credit: Volodymyr Nik/Shutterstock.com
Background
Due to their enhanced nanocomposite characteristics, environmental friendliness, and performance, biopolymer-based nanocomposites continue to draw interest for application as cutting-edge sustainable materials. For many different supramolecular architectures of heterogeneous hybrid systems, the layered double hydroxides (LDHs) are extensively used as carriers. Although high-performance micro-/nano-composite materials can be created by mixing nanofillers with polymers and biopolymers, some of these materials suffer from reduced oxidative degradation as a result of the nanoparticles.
It is possible that the additional nanofillers will hasten the oxidative breakdown of biopolymer matrices as well as certain polymers in particular circumstances. Unfortunately, this problem restricts the wide-ranging industrial use of nanocomposites.
Two distinct strategies have currently been put out in scientific literature to increase the stability of antioxidants and light stabilizers and prevent the stabilizers from becoming stuck between nanoceramic layers. Specifically, this involved the immobilization of stabilizing compounds through physical adsorption onto the nanoparticles on another surface and the anchoring of stabilizing molecules through chemical coupling onto particles and/or to polymer chains.
![Typical structure of nanoceramics, i.e., metal hydroxide layers and interlayer anions [6].](https://www.azom.com/images/news/ImageForNews_59839_1661414889562539.png)
Typical structure of nanoceramics, i.e., metal hydroxide layers and interlayer anions. Image Credit: Morici, E et al., Materials
About the Study
In this study, the authors formulated and characterized novel LDH-based nanocomposites, specifically Polyamide 11 (PA11). Two different hindered amine moieties, namely, HALS1 and HALS2, were anchored on the LDH layered internal structures and outer surfaces because LDH were thought to be carriers for hindered amine light stabilizing molecules.
Wide angle X-ray diffraction (WAXD), thermogravimetric studies, and spectroscopy supported the discovery of HALS1 and HALS2 in LDH. The novel LDH-HALS nanofillers were added to a PA11 matrix by the melt mixing at a rate of 5 wt.%. The resulting nanocomposites were then examined using rheological, morphological, and differential scanning calorimetric methods.
The team demonstrated that the LDH-HALS1/HALS2 nanofillers were perfectly disseminated into the PA11 matrix. Thin films exposed to ultraviolet B (UVB) were used to test the photo-oxidative resistance of PA11-based nanocomposite films, and the breakdown process was tracked over time by spectroscopic analysis. In comparison to PA11-based nanocomposites with unmodified LDH and the widely available hindered amine UV-stabilizer, the photo-oxidative resistance of the PA11/LDH-HALS1/HALS2 was tested.
It was determined that the PA11 nanocomposites were successfully shielded from UVB exposure by anchoring hindered amine moieties to the LDH. This was due to the hindered amine light stabilizing molecules being available to function in the crucial zone, which was at the interface between the inorganic and matrix particles where the degradation processes occurred.
The researchers used two distinct hindered amine light stabilizers as carriers for LDH, and the resulting nanofillers were then incorporated into a biopolymer matrix using melt mixing. The formulation of nanofillers with built-in stabilizing functionalities enabled the creation of nanocomposite films with better oxidation resistance. In addition to rheological and morphological analyses, differential scanning calorimetry was used to describe the synthesized nanocomposites. They were also exposed to accelerated UVB exposure to test their photo-oxidative resistance. It was discovered that anchoring the hindered amine moieties to the LDH effectively protected the nanocomposites against UVB exposure.
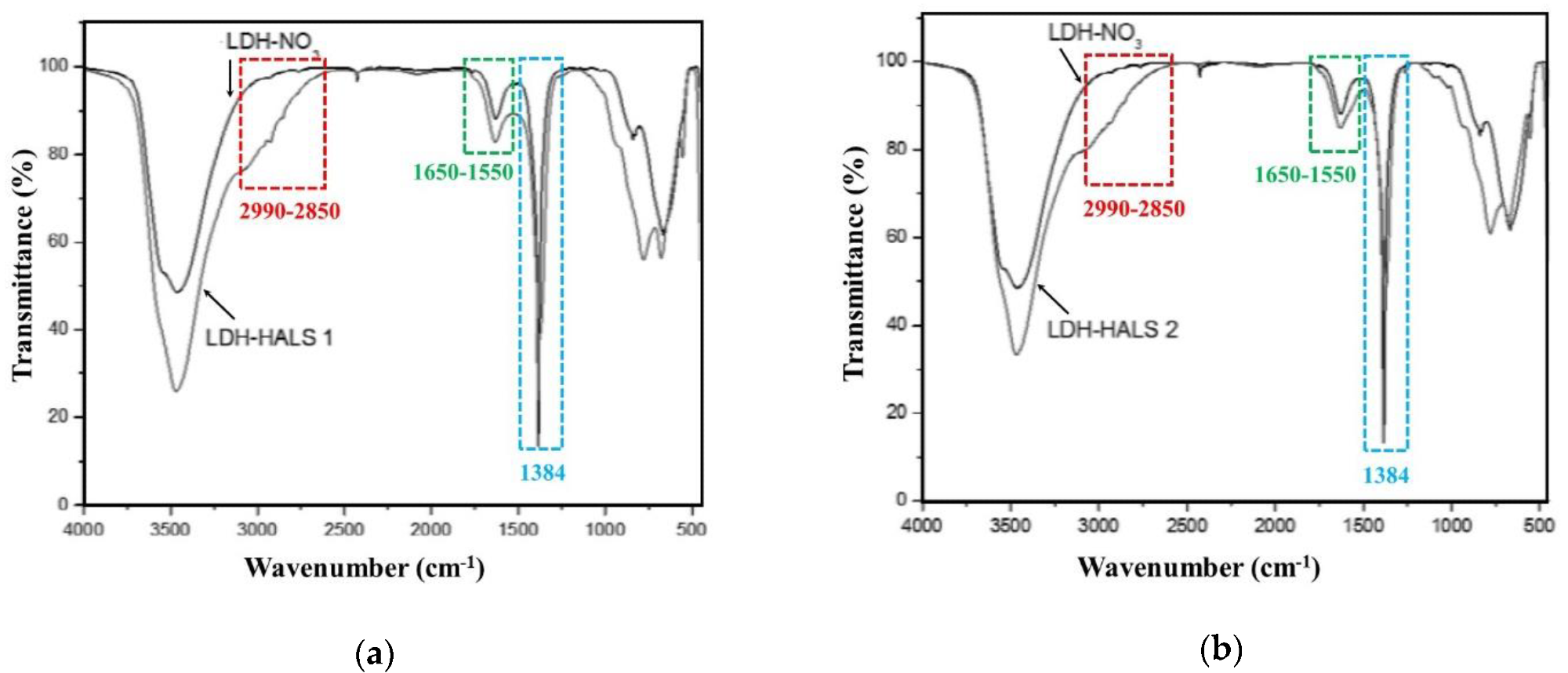
FTIR spectra of (a) LDH-HALS1 and (b) LDH-HALS2 in comparison to the spectrum of LDH-NO3.. Image Credit: Morici, E et al., Materials
Observations
Due to the presence of the γ-crystalline form, neat PA11 exhibited a shoulder at around 180 °C and a primary fusion peak at about 186.4 °C. The shoulder at around 180 °C was more noticeable when LDH was present, which indicated an increase in the PA11 γ-crystalline form. Both Tm1 and Tm2 were marginally lower due to the presence of LDH compared to the melting temperatures of neat PA11, and the outcomes were slightly better due to the presence of various LDH-HALS. When compared to clean PA11 at the same exposure duration, the addition of LDH in PA11 significantly increased the peak high to 1715 cm-1.
Due to ceramic-layered architectures, the PA11-based samples containing LDH-NO3 and LDH/HALS exhibited two clearly defined peaks, one broad peak at around 7° and one at approximately 11.2° in the range 2θ = 1.5-12°. Both LDH-HALS1 and LDH-HALS2 exhibited a three-stage decomposition process, with the first step occurring between 100 and 150 °C, the second between 180 and 250 °C due to the thermal decomposition of HALS1 and HALS2 molecules, and the third between 250 and 700 °C due to the dehydroxylation of the metal hydroxide layers and the loss of unexchanged nitrate ions.
The outcomes pointed to a positive impact from the presence of commercial HALS that was free-added and even more from HALS that was anchored. When present at the interface between inorganic particles and the host matrix or at the sites of degradation, the HALS molecules tethered to LDH had a distinctly discernible positive impact on the enhancement of the photo-oxidative resistance of biopolymer-based nanocomposite films.

SEM images at different magnifications of (a) PA11/LDH-NO3, (b) PA11/LDH-HALS1, (c) PA11/LDH-HALS2 and (d) PA11/LDH-HALSadded. Image Credit: Morici, E et al., Materials
Conclusions
In conclusion, this study created and characterized innovative, environmentally friendly, biopolymer-based nanocomposite films with LDH-HALS1 and LDH-HALS2 stabilization. Based on WAXD, FTIR, and TGA studies, the effective formulation of LDH-HALS2 and LDH-HALS1 was established. Melt mixing was used to create nanocomposites based on PA11 and both modified and unmodified LDH, which were then examined using rheological, morphological, and differential scanning calorimetry investigations. A sample based on PA11 that additionally contained free-added commercial HALS and LDH was created for comparison.
The authors mentioned that the LDH/HALS2 and LDH/HALS1 were spread and dispersed in a PA11 matrix very efficiently. In comparison to nanocomposites containing LDH and LDH/HALSadded, the photo-oxidative resistance of the PA11-based nanocomposites, including novel LDH-HALS1 and LDH-HALS2, was observed.
References
Morici, E., Infurna, G., Dintcheva, N. T., et al. Ecofriendly Biopolyester-Based Nanocomposite Films with Improved Photo-Oxidative Resistance. Materials, 15(16), 5778 (2022). https://www.mdpi.com/1996-1944/15/16/5778
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.