The extraordinary proton and oxide-ion (dual-ion) conductivities exhibited by the hexagonal perovskite-related oxide Ba7Nb3.8Mo1.2O20.1, as highlighted by researchers at Tokyo Tech, hold significant promise for upcoming electrochemical devices.
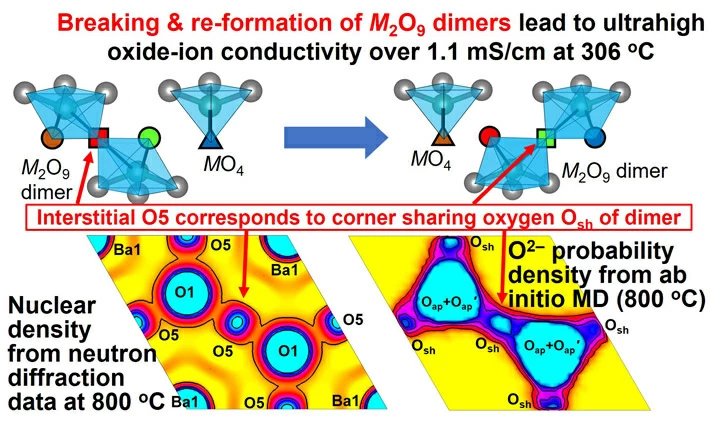 The top figure shows the snapshot for the oxide-ion migration. The red and green oxide ions move by breaking and reforming M2O9 dimers, which enables fast oxide-ion diffusion where the M cation is Nb5+ or Mo6+. The neutron scattering length density distribution from neutron diffraction data at 800 ℃ in the bottom left figure agrees with the time- and space-averaged probability density distribution of oxide ions from ab initio molecular dynamics simulations in the bottom right figure. The interstitial O5 atom in the bottom left figure corresponds to the corner-sharing oxygen atom (Osh in the bottom right figure and squares in the top figure). Image Credit: Tohoku University
The top figure shows the snapshot for the oxide-ion migration. The red and green oxide ions move by breaking and reforming M2O9 dimers, which enables fast oxide-ion diffusion where the M cation is Nb5+ or Mo6+. The neutron scattering length density distribution from neutron diffraction data at 800 ℃ in the bottom left figure agrees with the time- and space-averaged probability density distribution of oxide ions from ab initio molecular dynamics simulations in the bottom right figure. The interstitial O5 atom in the bottom left figure corresponds to the corner-sharing oxygen atom (Osh in the bottom right figure and squares in the top figure). Image Credit: Tohoku University
The elucidation of its distinctive ion-transport mechanisms is expected to lay the groundwork for superior dual-ion conductors. These advancements could prove pivotal in shaping the landscape of future clean energy technologies.
Clean energy technologies serve as the bedrock for sustainable societies, with solid-oxide fuel cells (SOFCs) and proton ceramic fuel cells (PCFCs) standing out as highly promising electrochemical devices for generating green power. Yet, these devices encounter hurdles that impede their advancement and widespread adoption.
Ideally, SOFCs should operate at lower temperatures to prevent detrimental chemical reactions that degrade their constituent materials. However, most known oxide-ion conductors crucial for SOFCs tend to display adequate ionic conductivity only at higher temperatures.
On the other hand, PCFCs face challenges—they are chemically unstable in carbon dioxide atmospheres and necessitate energy-intensive, high-temperature processing steps during manufacturing.
There exists a material type that holds the potential to address these challenges by merging the advantages of both SOFCs and PCFCs: dual-ion conductors. These materials, facilitating the movement of both protons and oxide ions, have the capacity to achieve elevated total conductivity levels at lower temperatures, thereby enhancing the efficiency of electrochemical devices.
While certain perovskite-related dual-ion conducting materials like Ba7Nb3.8Mo1.2O20.1 have been identified, their conductivities have not reached sufficiently high levels for practical applications, and their underlying conducting mechanisms remain insufficiently understood.
In light of these challenges, Professor Masatomo Yashima's research team at the Tokyo Institute of Technology, Japan, embarked on an exploration into the conductivity of materials akin to 7Nb4MoO20 but featuring a greater Mo fraction (specifically, Ba7Nb4-xMo1+xO20+x/2).
Their most recent study, conducted in collaboration with the Australian Nuclear Science and Technology Organisation (ANSTO), the High Energy Accelerator Research Organization (KEK), and Tohoku University, was published in Chemistry of Materials.
After screening different Ba7Nb4-xMo1+xO20+x/2 compositions, the researchers identified that Ba7Nb3.8Mo1.2O20.1 had extraordinary proton and oxide-ion conductivities.
Ba7Nb3.8Mo1.2O20.1 exhibited bulk conductivities of 11 mS/cm at 537 ℃ under wet air and 10 mS/cm at 593 ℃ under dry air. Total direct current conductivity at 400 ℃ in wet air of Ba7Nb3.8Mo1.2O20.1 was 13 times higher than that of Ba7Nb4MoO20, and the bulk conductivity in dry air at 306 ℃ is 175 times higher than that of the conventional yttria-stabilized zirconia (YSZ).
Masatomo Yashima, Professor, Tokyo Institute of Technology
The researchers aimed to unveil the fundamental mechanisms contributing to these elevated conductivity levels. Employing ab initio molecular dynamics (AIMD) simulations, neutron diffraction experiments, and analyses of neutron scattering length density, they delved deeper into the structure of Ba7Nb3.8Mo1.2O20.1. These sophisticated techniques allowed for a more comprehensive understanding of what distinguishes this material as a dual-ion conductor.
The team uncovered that the heightened oxide-ion conductivity in Ba7Nb3.8Mo1.2O20.1 stems from a distinctive phenomenon. It appears that adjacent MO5 monomers within Ba7Nb3.8Mo1.2O20.1 can create M2O9 dimers by sharing a corner oxygen atom (M represents the Nb or Mo cation).
The disassembly and reformation of these dimers result in the swift movement of oxide ions, akin to a continuous line of individuals passing buckets of water along. Additionally, the AIMD simulations unveiled that the observed heightened proton conduction occurs due to efficient proton migration within the hexagonally close-packed BaO3 layers present in the material.
The findings of this study underscore the potential of perovskite-related dual-ion conductors and offer insights that could serve as blueprints for the strategic development of such materials.
The present findings of high conductivities and unique ion migration mechanisms in Ba7Nb3.8Mo1.2O20.1 will help the development of science and engineering of oxide-ion, proton, and dual-ion conductors.
Masatomo Yashima, Professor, Tokyo Institute of Technology
Journal Reference:
Sakuda, Y., et al. (2023). Dimer-Mediated Cooperative Mechanism of Ultrafast-Ion Conduction in Hexagonal Perovskite-Related Oxides. Chemistry of Materials. doi.org/10.1021/acs.chemmater.3c02378.