Mar 4 2016
A method to synthesize the first binary compound of oxygen and krypton - a krypton oxide, has been formulated by a team of theoretical chemists at the Institute of Physical Chemistry of the Polish Academy of Sciences. It has been discovered that this unusual substance can be produced even in the existing laboratories using very high pressure.
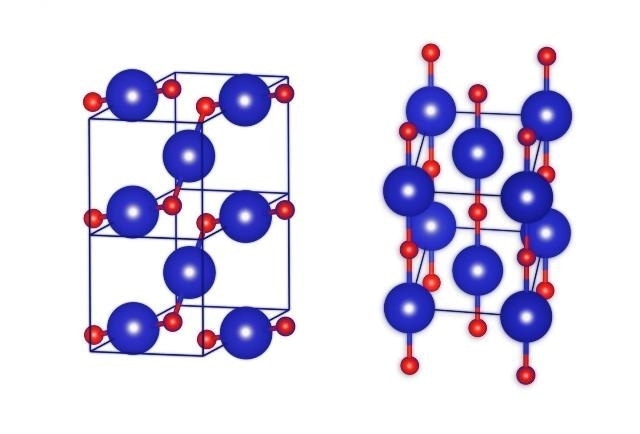 This image shows the crystal structures of krypton monoxide KrO: more stable on the left, less stable on the right. Krypton atoms coloured blue, oxygen atoms coloured red. (Photo credit: IPC PAS)
This image shows the crystal structures of krypton monoxide KrO: more stable on the left, less stable on the right. Krypton atoms coloured blue, oxygen atoms coloured red. (Photo credit: IPC PAS)
Superman and his race considered crystals of kryptonite to be deadly. The material was thought to be created on the planet Krypton, and was likely to be created under extremely high pressure.
Real krypton’s progenitor is an element with an atomic number of 36, a noble gas that was not able to form stable chemical compounds.
Two theoretical chemists, from the Institute of Physical Chemistry of the Polish Academy of Sciences (IPC PAS) in Warsaw, Poland, presented their research findings in the Scientific Reports journal. The article describes a method to produce a unique crystalline material, where krypton atoms can be chemically bonded to another element.
The substance we are predicting is a compound of krypton with not nitrogen, but oxygen. In the convention of the comic book it should, therefore, be called not so much kryptonite as kryptoxide. So if Superman's reading this, he can stay calm - at the moment there's no cause for panic! Our krypton monoxide, KrO, probably does not exist in nature. According to current knowledge, deep in the interiors of planets, that is, the only place where there is sufficient pressure for its synthesis, oxygen does not exist, nor even more so, does krypton.
Dr. Patrick Zaleski-Ejgierd, IPC PAS
Earlier under cryogenic conditions, compounds of krypton were created in a laboratory. However they were merely single, linear, and tiny molecules of the hydrogen-carbon-krypton-carbon-hydrogen type.
The Polish team speculated whether krypton that bonded chemically with another element, could also be capable of forming a broad and stable crystal lattice when exposed to certain conditions. The researchers used genetic algorithms and models based on a “density functional theory”. Typically, in solid state physics this theory has been a standard tool for the research and description of the whole spectrum of chemical molecules.
Our computer simulations suggest that crystals of krypton monoxide will be formed at a pressure in the range of 300 to 500 million atmospheres. This is a huge pressure, but it can be achieved even in today's laboratories, by skillfully squeezing samples in diamond anvils.
Pawe Lata, PhD Student, IPC PAS
Crystal lattices are produced using molecules or atoms arranged in space in a logical manner. The smallest repetitive piece of these structures is called a unit cell. It is the basic 'building block'. For instance, the unit cell of crystals of table salt has a cuboid-shape, where chlorine and atoms, arranged one after the other, are mounted on each corner, so close to each other that they are bound by covalent chemical bonds.
The unit cell of krypton monoxide is in the shape of a cube with a diamond base, with the krypton atoms located at the corners. A single atom of krypton can be found in the middle of the two opposite side walls.
Where is the oxygen? On the side walls of the unit cell, where there are five atoms of krypton, they are arranged like the dots on a dice showing the number five. Single atoms of oxygen are located between the krypton atoms, but only along the diagonal - and only along one! Thus, on each wall with five krypton atoms there are only two atoms of oxygen. Not only that, the oxygen is not exactly on the diagonal: one of the atoms is slightly offset from it in one direction and the other atom in the other direction.
Pawe Lata, PhD Student, IPC PAS
Each oxygen atom is chemically bound to the two nearest adjoining atoms of krypton in this highly distinctive unit cell. Long polymer structures are formed when zigzag chains of Kr/O\Kr\O/Kr travel via the crystal of krypton monoxide. Crystals of this type of krypton monoxide are said to possess properties of a semiconductor based on certain calculations. The crystals are likely to be dark with poor transparency.
The researchers at IPC PAS have discovered another compound of krypton - tetroxide KrO4, which is a little less stable. This compound possesses a simpler crystalline structure, and could be produced at a pressure of more than 340 million atmospheres. It probably possesses characteristics of a metal.
Once these two types of krypton oxide crystals are formed, they could possibly exist at a fairly lower pressure than that required during their formation. However, the pressure on earth is quite low, which would mean that the crystals would be decomposed instantly.
Reactions occurring at extremely high pressure are almost unknown, very, very exotic chemistry. We call it 'Chemistry on the Edge'". Often the pressures needed to perform syntheses are so gigantic that at present there is no point in trying to produce them in laboratories. In those cases even methods of theoretic description fail! But what is most interesting here is the non-intuitiveness. From the very first to the last step of synthesis you never know what's going to happen.
Dr. Patrick Zaleski-Ejgierd, IPC PAS
This study was funded by an OPUS grant from the Polish National Science Centre.