Aug 3 2013
A team of Penn State researchers at the Applied Research Laboratory (ARL) and Materials Science Department have seen the Left Ventricular Assist Device (LVAD) they developed between 2005 and 2011 transitioned to its first successful human test in March.
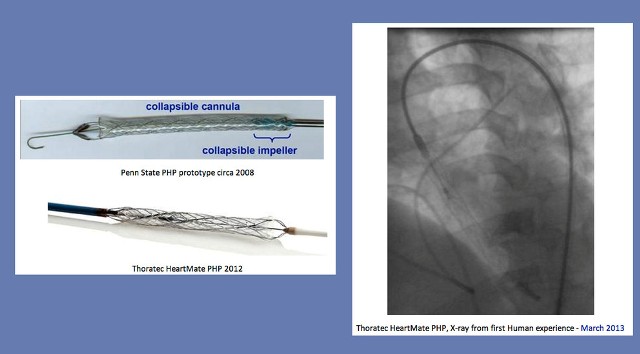 Images of the percutaneous heart pump. Image: Penn State
Images of the percutaneous heart pump. Image: Penn State
Thoratec Corp., a world leader in mechanical circulatory support therapies for failing hearts, licensed the technology from Penn State in 2010. LVADs are designed to partially take over the pumping function of a failing heart. In less than three years, the preliminary design components, test facilities and manufacturing techniques developed at Penn State to build a prototype device have been evolved by Thoratec to where the LVAD could be tested in humans.
“The excitement for our Penn State team is that our research is now well on its way to saving lives,” said Rob Kunz, ARL researcher and principal investigator on the project. “The design goals for the now-termed HeartMate PHP (percutaneous heart pump) were daunting: Develop a blood pump on the end of a long, thin catheter small enough to be inserted through an incision in a patient’s thigh and snaked up through the descending aorta to the left ventricle to be deployed. The PHP has to provide enough blood-pumping capability to support an adult male (approximately 4.5 liters per minute at a 60mmHg pressure head) for several days, without causing undo damage to the heart or the blood itself.”
Many commercially available long-term LVADs require that the chest wall be surgically opened for device insertion, a complicated and traumatic procedure. The design goals for the short-term PHP device were motivated by a desire for a rapidly inserted minimally invasive acute support technology. The PHP is designed to be inserted, deployed and operating within minutes of a patient suffering a heart attack or other acute heart condition, potentially prolonging the life of tens of thousands of people in the United States each year.
With these requirements, industry came to Penn State because of the University’s strong capabilities in hydrodynamics (design, testing and modeling) and materials science, both critical technologies in any blood pump application. “We were essentially given the requirements and a blank sheet of paper,” said Kunz, “It did not take long before we had developed the basic concept: a very small, very rapidly spinning collapsible impeller, housed within a collapsible cannula."
Developing the concept for this tiny but efficient turbomachine has required the Penn State team to overcome multiple challenges. An important one has to do with the nature of the fluid. The LVAD works by means of an impeller that pumps blood while spinning at approximately 19,000 RPM; but you can’t put blood in a blender – even a tiny one – and expect it to maintain the physical and biological characteristics that enable it to carry oxygen. Instead, the impeller blades had to be engineered so that they minimally damage the red blood cells.
Another major challenge was that the material from which the LVAD blades are made had to be flexible enough to compress within a 4-mm catheter. That same elasticity means the blades deflect under the fluid load when they spin. Thus, the blades had to be designed to offset the deflection while at the same time preserving their desired hydrodynamic performance. Thirdly, the cannula in which the impeller spins, and which guides the blood flow through the pump, also had to be collapsible – essentially a covered stent with enough integrity to support the pumping while being flexible enough to fold completely into a 4-mm catheter.
Said Kunz, “This has been a truly multidisciplinary design and development effort, involving experts in materials science, experimental fluid mechanics, computational fluid mechanics, engineering design, bearing technology, rapid prototyping and controls, as well as machinists and integration testing technicians. We also had the exciting opportunity to become involved in several technologies well outside of our core competencies including circulatory system physiology, blood cell mechanics/biochemistry, animal testing and materials that are ‘cleared’ for use in the body. Also, despite its ‘design & deliver’ focus, the project supported a Penn State Ph.D. thesis in Mechanical Engineering on the topic of fluid-structure interaction modeling.”
“So where is the device now and where is it going?” Kunz continued, “Four PHP-related patents have been issued with members of our team as co-inventors. The Penn State technology management office successfully negotiated licensing agreements with Thoratec, so the successful human test gave rise to Penn State’s first licensing revenue from the device.”
Thoratec is a great transition partner for this technology as they are one of only a handful of companies in the world that have the expertise and resources to mature blood pump devices though manufacturing development and clinical trials to commercialization. Thoratec intends to continue clinical trials on the use of the HeartMate PHP to support patients undergoing high-risk percutaneous interventions, as well as those in cardiogenic shock, a population with persistently high mortality rates. The company anticipates initiating a European study in the second half of 2013, followed by a U.S. clinical trial in early 2014.