Reviewed by Mila PereraSep 15 2022
The demand for high-density batteries grows with the extensive adoption of alternative energy sources and electric cars; therefore, enhancing the stability and volume of lithium-ion batteries is a major requirement.
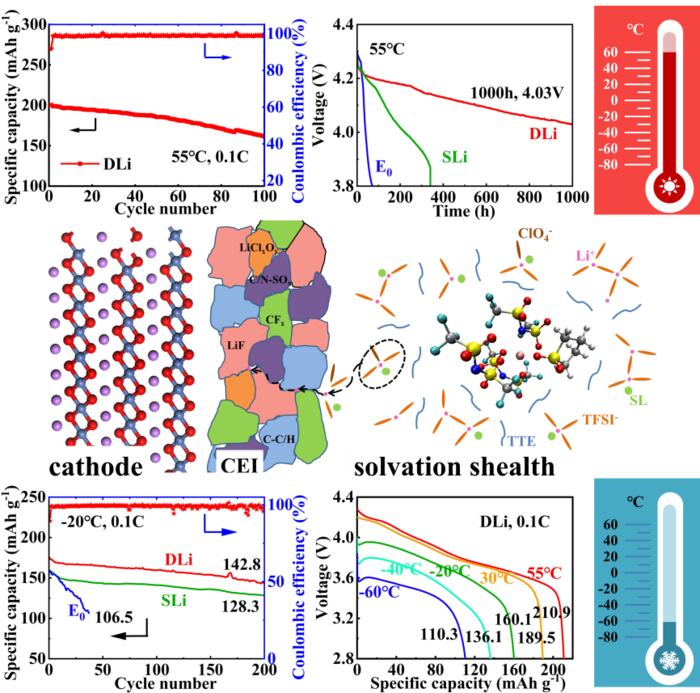 The diagrams in this picture show the makeup of the cathode, how the cathode and the electrolyte interact (CEI), and the solvation sheath, which is how the different chemical molecules in the solvent interact with each other. The graphs show how the electrolyte responded at high and low temperatures, including a graph for showing capacity over time and capacity at different voltages. Image Credit: Nano Research
The diagrams in this picture show the makeup of the cathode, how the cathode and the electrolyte interact (CEI), and the solvation sheath, which is how the different chemical molecules in the solvent interact with each other. The graphs show how the electrolyte responded at high and low temperatures, including a graph for showing capacity over time and capacity at different voltages. Image Credit: Nano Research
Modern lithium-ion battery technology, which regularly uses nickel, is less stable at very high temperatures, resulting in overheating due to high voltages and temperature and making them prone to weaken rapidly.
To resolve this issue, scientists are exploring new chemical combinations that can remedy these shortcomings. In a recent study, researchers showed how an inorganic compound additive and a solvent could enhance the performance and stability of lithium-ion batteries with nickel cathodes.
The results of their study have been published in the September 12th, 2022, issue of the journal Nano Research.
The fundamentals of how batteries function are the same for an average household AA battery and an industrial lithium-ion battery. The cathode forms the positive electrode, the anode forms the negative electrode, and the electrolyte is the solution between them inside the battery.
Positively and negatively charged ions travel through the electrolyte, and a chemical reaction produces electrical energy. In this study, scientists discovered that a sulfolane-based liquid electrolyte with the addition of lithium perchlorate works as a probable solution to typical lithium-ion battery shortcomings.
For nickel-based cathodes, good low-temperature electrochemical performance is usually achieved at the expense of property and safety at room temperatures. This is because electrolytes with low melting solvents deteriorate dramatically. The high volatility and flammability of these electrolytes also limit their application at high temperatures.
Fang Lian, Professor, School of Materials Science and Engineering, University of Science and Technology Beijing
The team learned they could tackle many of these shortcomings by incorporating lithium perchlorate into the sulfolane.
Sulfolane is a solvent that was first developed for use in the oil and gas sector, but it is currently employed in several different industrial locations because it remains stable at very high temperatures.
When incorporated into sulfolane, lithium perchlorate, an inorganic compound, helps maintain the electrolyte's stability. A third chemical is incorporated to dilute the electrolyte and support the stability of the electrolyte at a broad range of temperatures.
To test if the recommended electrolyte functioned efficiently, the team developed a battery using the electrolyte before performing a series of tests and theoretical calculations. They learned that the solvent could preserve conductivity at a broad range of temperatures, ranging from -60 °C to 55 °C.
In contrast, conventional electrolytes tend to solidify at temperatures less than -20 °C. Incorporating lithium perchlorate into the electrolyte reinforces how the various electrolyte chemicals interact with each other and decrease the amount of energy needed, rendering it easier for the electrolyte to function at lower temperatures.
The diluted high concentration sulfolane-based electrolyte with lithium perchlorate additive realizes the wide-temperature application in high voltage cells. This combination improves the lithium-ion transference and reduces the desolvation energy, while inhibiting the continuous decomposition of the electrolyte and the acute deterioration of the cathode at high temperatures.
Fang Lian, Professor, School of Materials Science and Engineering, University of Science and Technology Beijing
“Our work provides a comprehensive understanding of the molecular design of the electrolyte, facilitating the development of high energy density lithium batteries,” Lian added.
Other study contributors include Shuang He, Yixin Zhu, and Jiayi Ding of the School of Materials Science and Engineering at the University of Science and Technology Beijing and Guangyu Zhao at the University of New South Wales.
The National Key R&D Program of China (Grant No. 2018YFB0104300) and the National Natural Science Foundation of China (Grant Nos. 52172180, 51872026) supported this study.
Journal Reference
Zhu, Y., et al. (2022) The sulfolane-based liquid electrolyte with LiClO4 additive for the wide-temperature operating high nickel ternary cathode. Nano Research. doi.org/ 10.1007/s12274-022-4852-y.