Reviewed by Mila PereraSep 9 2022
Scientists at the U.S. Department of Energy’s (DOE) Argonne National Laboratory (ANL) are known for their past revolutionary discoveries with lithium-ion (Li-ion) batteries. Most of these discoveries have concentrated on a battery cathode referred to as NMC, a nickel-manganese-cobalt oxide. Batteries possessing this cathode currently power the Chevy Bolt.
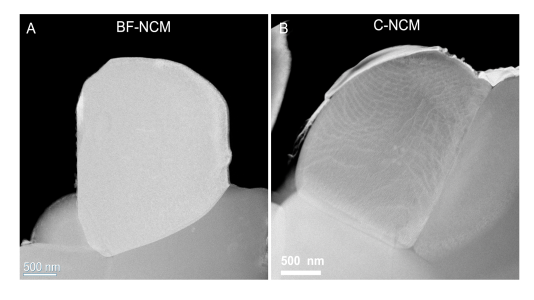 Image shows single crystals of cathode material: (A) no internal boundaries and (B) internal boundaries visible. Image Credit: Argonne National Laboratory.
Image shows single crystals of cathode material: (A) no internal boundaries and (B) internal boundaries visible. Image Credit: Argonne National Laboratory.
The scientists at Argonne have made yet another innovation with the NMC cathode. A new structure has been created for the cathode’s micro-sized particles, leading to safer and longer-lasting batteries capable of working at high voltage and power vehicles to cover greater driving distances.
The present-day NMC cathode has posed a major barrier to operation at high voltage.
Guiliang Xu, Assistant Chemist, Argonne National Laboratory
With the charge-discharge cycling process, performance quickly drops due to cracks developing in the cathode particles. For over a few decades, battery scientists have been looking for ways to remove these cracks.
One previous method involved microscale spherical particles comprising several much smaller particles. The large spherical particles are polycrystalline, with crystalline regions oriented differently. Consequently, they have what is referred to as grain boundaries between particles by researchers, which cause cracking during battery cycling.
To avoid this, Xu and Argonne's team earlier created a polymer coating as a shield around each particle. This coating encompasses the large spherical particles and smaller particles within them.
A diverse method to stop this cracking requires single-crystal particles. Electron microscopy of these particles showed that they possess no boundaries.
The issue facing the team was that cathodes composed of both coated polycrystals and single crystals still developed cracks with cycling. Therefore, they exposed these cathode materials to substantial analyses at the Advanced Photon Source (APS) and Center for Nanoscale Materials (CNM), DOE Office of Science user facilities at Argonne.
A number of X-Ray analyses were performed at five APS beamlines (11-BM, 20-BM, 2-ID-D, 11-ID-C, and 34-ID-E). As demonstrated by electron and X-ray microscopy, the outcome of the tests revealed to the researchers that the single crystals essentially had boundaries inside. Scanning and transmission electron microscopies at CNM validated the finding.
When we look at the surface morphology of these particles, they look like single crystals. But when we use a technique called synchrotron X-ray diffraction microscopy and other techniques at the APS, we find boundaries hiding inside.
Wenjun Liu, Physicist, Argonne National Laboratory
Significantly, the researchers formulated a technique for creating boundary-free single crystals. Analyzing small cells with such single-crystal cathodes at extremely high voltage revealed a 25% boost in energy storage per unit volume, with virtually no drop in performance throughout 100 cycles of testing.
Comparatively, across the same cycle life, the capacity dropped from 88% to 60% in NMC cathodes made up of single crystals with several internal boundaries or with coated polycrystals.
Atomic scale calculations exposed the mechanism underlying the capacity drop in the cathode. According to nanoscientist Maria Chan in CNM, in contrast to the regions away from them, boundaries are more susceptible to losing oxygen atoms while the battery is charging. This oxygen loss results in degradation with cell cycling.
Our calculations showed how boundaries lead to oxygen release at high voltage and, therefore, performance decline.
Maria Chan, Nanoscientist, Argonne National Laboratory
Removing the boundaries stops the release of oxygen, thus increasing the safety of the cathode and stability with cycling. Oxygen release measurements at APS and the Advanced Light Source at DOE’s Lawrence Berkeley National Laboratory validated this finding.
Khalil Amine, a Distinguished Fellow at the Argonne National Laboratory stated, "We now have guidelines that battery manufacturers can use to prepare cathode material that is boundary free and works at high voltage. And the guidelines should apply to other cathode materials besides NMC."
The research paper was published in the journal Nature Energy. Besides Xu, Amine, Liu, and Chan, Argonne authors include Xiang Liu, Venkata Surya Chaitanya Kolluru, Chen Zhao, Xinwei Zhou, Yuzi Liu, Liang Yin, Amine Daali, Yang Ren, Wenqian Xu, Junjing Deng, Inhui Hwang, Chengjun Sun, Tao Zhou, Ming Du, and Zonghai Chen.
Others involved in this study were researchers from Lawrence Berkeley National Laboratory (Wanli Yang, Qingtian Li, and Zengqing Zhuo), Xiamen University (Jing-Jing Fan, Ling Huang, and Shi-Gang Sun), and Tsinghua University (Dongsheng Ren, Xuning Feng, and Minggao Ouyang).
The study received support from the DOE Vehicle Technologies Office.
Journal Reference:
Liu, X., et al. (2022) Origin and regulation of oxygen redox instability in high-voltage battery cathodes.Nature Energy. doi.org/10.1038/s41560-022-01036-3.