Feb 13 2015
A research team from the Harvard’s School of Engineering and Applied Sciences (SEAS) and Wyss Institute for Biologically Inspired Engineering has carried out a new study on a powerful, long-lasting repellent surface technology for use with medical materials to avert infections due to biofilms.
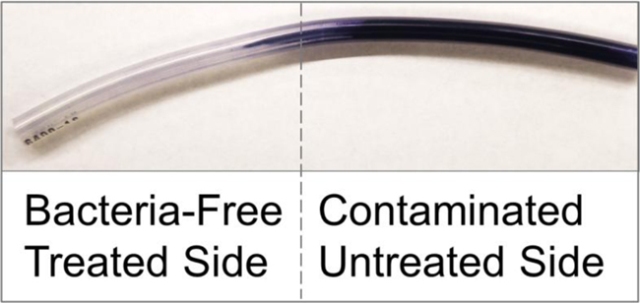 Harvard researchers have demonstrated a powerful, long-lasting repellent surface technology that can be used with medical materials to prevent infections caused by biofilms. (Credit. Joanna Aizenberg.)
Harvard researchers have demonstrated a powerful, long-lasting repellent surface technology that can be used with medical materials to prevent infections caused by biofilms. (Credit. Joanna Aizenberg.)
Bacterial build-up in the human body one of the most typical causes of microbial infections. Accumulation of bacterial cells in the body results in the formation of adhesive colonies called biofilms. These biofilms enable the bacteria to thrive in the body leading to several infections and life-threatening risks to their hosts. The main site of formation of biofilms is on the medical surfaces such as implants, intravenous catheters, urinary catheters and mechanical heart valves.
The new technology employs liquid-infused polymers capable of storing lubricating liquids in significant quantities, in their sponge-like molecular structure. This feature enables absorption of large amount of lubricant that travels to the surface making it slippery and impervious. As a result, the environment becomes unsusceptible to the colonization of bacteria.
The team is led by Joanna Aizenberg - the Amy Smith Berylson Professor of Materials Science at Harvard SEAS, Co-Director of the Kavli Institute for Bionano Science and Technology, Wyss Institute Core Faculty member and Professor of Chemistry and Chemical Biology in Harvard’s Faculty of Arts and Sciences.
The researcher chose a solid silicone polymer for this study as a similar material has been already used in the medical tubing that is saturated with a silicone oil liquid. Both the components are harmless and free of toxic substances, making them ideal for applications in medical devices and cosmetics.
The solid silicone tubing is saturated with silicone oil, soaking it up into all of the tiny spaces in its molecular structure so that the two materials really become completely integrated into one.
Caitlin Howell - Postdoctoral Researcher / Co-author
Wyss Institute
This saturation process improves the efficiency of the liquid-infused polymer resulting in a material that can hold out against conventional sterilization methods. This is possible because of the continuous diffusion of silicon oil to the surface preventing the loss of slipperiness from the surface. By doing so, the surface replenishes itself to compensate the removal of oil by flowing liquids such as gastro-intestinal fluids, blood and urine, inside the body.
Noah MacCallum, an exchange undergraduate student at SEAS and the lead author of the study exposed medical tubing, both treated and untreated to common pathogens like Staphylococcus epidermidis, Escherichia coli and Pseudomonas aeruginosa which form biofilms. The experimental findings suggested that the liquid-infused polymer tubing prevented the formation of biofilm and minimized bacterial adhesion.
This new approach can also be used for preventing bacterial infections due to the biofilm formation on catheters and similar medical devices. Removal of biofilm is a critical part as these biofilms, once formed, cannot be easily cleared, inducing antibiotic resistance in bacteria.
“With widespread antibiotic resistance cropping up in many strains of infection-causing bacteria, developing out-of-the-box strategies to protect patients from bacterial biofilms has become a critical focus area for clinical researchers. Liquid-infused polymers could be used to prevent biofilms from ever taking hold, potentially reducing rates of infection and therefore reducing dependence on antibiotic use," said Wyss Institute Founding Director Donald Ingber, who is also the Judah Folkman Professor of Vascular Biology at Harvard Medical School and Boston Children’s Hospital and Professor of Bioengineering at Harvard SEAS.
Aizenberg and her team earlier developed several slippery surface technologies such as the Slippery Liquid-Infused Porous Surfaces (SLIPS) technology in 2011, which is capable of repelling any solid or liquid material, providing a wide range of applications.
SLIPS served as a basis for the development of another type of slippery surface that uses tethered-liquid perfluorocarbons called TLP coating. Developed by Aizenberg and Ingber together in 2014, the TLP coating make use of FDA-approved materials in order to prevent the occurrence of sepsis and bacterial formation, without using conventional anticoagulant drugs prone to side effects.
We could apply liquid-infused polymers to other materials plagued with biofouling problems, such as waste-water management systems, maritime vessels or oil pipes.
Philseok Kim - Former Senior Research Scientist / Co-author / Vice President
Wyss Institute / SLIPS Technologies
With the successful experimental findings, Aizenberg and the team believe that this new approach will further strengthen the advancements in slippery surface technologies.
“Each technology in our portfolio has different properties and potential uses, but collectively this range of approaches to surface coatings can prevent a broad range of life-threatening problems, from ice accumulation on airplane wings to bacterial infections in the human body,” said Aizenberg.