Oct 18 2016
 Researchers at Princeton have developed a two-component catalyst system that works in tandem to selectively activate the strongest bond in the molecule, a nitrogen-hydrogen (N-H) bond, through a process known as proton-coupled electron transfer (PCET). Credit: Knowles lab
Researchers at Princeton have developed a two-component catalyst system that works in tandem to selectively activate the strongest bond in the molecule, a nitrogen-hydrogen (N-H) bond, through a process known as proton-coupled electron transfer (PCET). Credit: Knowles lab
Princeton University researchers have developed an innovative method to develop valuable carbon-carbon bonds using ubiquitous but conventionally unreactive carbon-hydrogen bonds. The details of the research have been published in Nature.
The technique provides chemists with a new path to construct carbon-carbon bonds that bypass the requirement for reactive functional groups. Efficient carbon-carbon bond forming reactions are highly in demand and vital for preparing molecular complexity in industrial materials and pharmaceuticals.
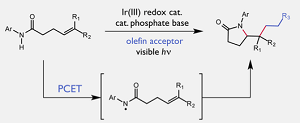
The reaction was enabled by a method called as proton-coupled electron transfer (PCET), which is proven in the inorganic field but not fully explored in synthetic settings.
We're trying to broaden organic synthesis by pulling from inorganic chemistry and biology and bringing in new mechanisms that people can consider when they're designing reactions.
Gilbert Choi, Graduate Student, Princeton University
In the last few years, research conducted in the Knowles lab has been dedicated to comprehending how PCET can be manipulated in organic reactions. The basic idea centers on using two autonomous catalysts - an oxidant and base - to homolyze, or divide in half, strong chemical bonds. In this study, the team aimed to achieve the strongest bond yet, the nitrogen-hydrogen bond in compounds known as N-alkyl amides.
We've been able to homolyze all types of bonds, but this particular reaction was exciting because amide N-H bonds are exceptionally strong and cannot be homolyzed using conventional approaches.
Robert Knowles, Assistant Professor of Chemistry, Princeton University
The team used a phosphate base and a light-activated iridium-catalyst to divide the nitrogen-hydrogen bond and produce an important radical intermediate. The intermediate consecutively breaks a weaker carbon-hydrogen bond present, which then turns into the site for the development of a new carbon-carbon bond.
The Princeton team successfully showed that their catalyst system functioned for a number of intramolecular reactions, meaning that the initial amide and ensuing carbon-carbon bond are linked to the same molecule, as well as the more difficult intermolecular reactions, where the new bond and the amide are separate.
This reaction represents a novel route for reactivity and paves the way for several new synthetic opportunities, Knowles said.
We think these methods will enable the development of a wide range of new C-H functionalization technologies.
Robert Knowles, Assistant Professor of Chemistry, Princeton University